By Irina Britva, Ph.D and Jennifer Chheda, Ph.D.
On February 8, 2017, the PTAB denied Fustibal LLC’s (“Fustibal”) petition to institute inter partes review of U.S. Patent 8,637,553 B2 (“the ’553 patent”) owned by Bayer HealthCare LLC (“Bayer”) (IPR2016-01490). The 553 patent has also been asserted in two district court proceedings against Teva Pharmaceuticals USA, Inc. and Apotex, Inc. In addition, the development of the compound covered by the claims of the 553 patent is the subject of an ongoing litigation with Onyx Pharmaceuticals, Inc.
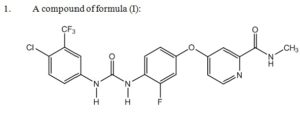
The ’553 patent relates to regorafenib (formula (I) below) which is the active ingredient in the anti-cancer drug commercialized by Bayer as STIVARGA® for use in treating certain types of colorectal cancer. Representative claim 13 of the ’553 patent recites:
In its petition for inter partes review (“IPR”) of the 553 patent, Fustibal asserted that all of the claims in ‘553 patent are unpatentable on grounds of anticipation and obviousness. In particular, Fustibal alleged that the claims of the 553 patent are anticipated by a primary prior art reference disclosing a class of compounds of a certain general formula useful for the treatment of cancer as well as numerous examples of compounds within the scope of the formula, including the formula for sorafenib and three examples of sorafenib with certain substitutions. Fustibal also alleged that all of the claims of the ‘553 patent were obvious over this primary prior art reference alone or in combination with certain secondary prior art references.
On the ground of anticipation, Fustibal, relying on its expert’s declaration, alleged that the primary prior art reference expressly discloses regorafenib because the reference discloses a sufficiently small genus of compounds and that small genus would be readily apparent to one of skill in the art who was looking to modify sorafenib. In response, Bayer argued that the primary prior art reference was considered by the Examiner as the closest prior art and was overcome during prosecution. In addition, Bayer argued that Fustibal’s reasoning is legally insufficient to establish anticipation because “any desire to modify sorafenib relates to obviousness, not anticipation”. See Fustibal LLC v. Bayer HealthCare LLC, IPR2016-01490 (PTAB February 8, 2017), Paper 9 at 15.
The PTAB agreed with Bayer that the primary prior art reference was considered during prosecution of the application and that the claims of the ‘553 patent were expressly allowed over the primary prior art reference. Thus, the PTAB declined to institute an IPR on the ground of anticipation. In addition, the PTAB declined to institute an IPR on the ground of anticipation on the merits because Fustibal failed to establish a reasonable likelihood that the claims of the ‘553 patent are anticipated by the primary prior art reference. In particular, the PTAB found that Fustibal had failed to demonstrate that one of ordinary skill in the art would find that the primary prior art reference disclosed a genus sufficiently small so as to anticipate the claims of the ‘553 patent. The PTAB agreed with Bayer that the small genus described by Fustibal does not exist in the primary prior art reference and only results from Fustibal’s “improper picking and choosing disparate aspects of the disclosure.” Fustibal LLC, IPR2016-01490, Paper 9 at 14.
On the grounds of obviousness, the PTAB again declined to institute an IPR because the primary prior art reference was considered and overcome during prosecution of the application issuing as the ‘553 patent. In addition, the PTAB declined to institute an IPR on the merits of the alleged grounds for obviousness. The PTAB agreed with Bayer that Fustibal did not engage in the two prong inquiry for determining whether a claimed compound would have been obvious over prior art compounds. In particular, the PTAB found that Fustibal failed to provide an explanation as to why the person of ordinary skill in art would have selected sorafenib as the starting point for further modification. In addition, the PTAB found that Fustibal had failed to provide an adequate explanation as to why a person of ordinary skill in the art would have had a reason to modify sorafenib to make the claimed compound with a reasonable expectation of success. Accordingly, the PTAB concluded that Fustibal had not established a reasonable likelihood that the claims of the ‘553 patent are rendered obvious by the primary prior art reference alone or in combination with the secondary prior art references.
Takeaway
This decision demonstrates the difficulty that may be encountered when asserting unpatentability on the grounds of anticipation and obviousness in an IPR over a reference considered during prosecution of the application issuing as the patent. In addition, the decision emphasizes the importance of carefully articulating the bases for establishing obviousness of a claimed compound based upon prior art compounds.
Latest posts by Cary Miller (see all)
- IPR Estoppel Applies Even After A Bench Trial On Validity - May 2, 2019
- Communications with, and Criticism by, FDA in Analyzing Obviousness - April 30, 2019
- IPR Appeal Dismissed After Biosimilar Development Discontinued - February 14, 2019